A future focused company targeting solutions aimed at delivering a positive global impact
Link to: Soil Carbon Sequestration & Nutrient Management
Soil Carbon Sequestration & Nutrient Management
We are focused on sustainability and circularity
Our team is empowered to collaborate and innovate, utilising proprietary technology and know-how to develop advanced materials and deliver solutions aimed at addressing sustainability challenges.
We are developing advanced materials
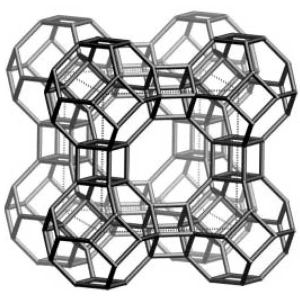
Zeolites are aluminosilicate materials with a cage-like structure, that link into frameworks and can be designed to selectively adsorb molecules or ions dependent on their unique construction.
We have an experienced team
and established
external networks
We have a highly skilled team with deep experience in research and development and a strong track record in project delivery and commercialisation.